Human Testing--The Lab Rat Myth
Chang Chiung-fang / photos Chuang Kung-ju / tr. by Scott Williams
January 2008

Human testing has been a neces-sary, if risky, aspect of medical research since those distant days when the mythical emperor Shennong, the legendary founder of Chinese agriculture and medicine, sampled hundreds of plants in his search for herbal remedies. But history also abounds in examples of inhumane and unethical quacks who sacrificed their subjects in their research.
Must the relationship between medical advances and human rights be antagonistic? Are there criteria for making a choice when research interests collide with human rights?
An answer can be found in the 1964 Helsinki Declaration on biomedical research, which proclaims: "Concern for the interests of the subject must always prevail over the interests of science and society."
However, though the principle is clear, it's not always honored.
Example 1: In November 2007, a former head of the Department of Anesthesiology at the Kaohsiung branch of Chang Gung Memorial Hospital came under fire from the medical community for having conducted human experiments without the permission of the hospital or the consent of his patients. The doctor injected Midazolam, a sedative approved only for intramuscular or intravenous injection, into the spines of 30 diabetic patients undergoing surgery. The doctor went on to boast in his published results that the use of a spinal block consisting of low doses of Bupivacaine together with Midazolam on diabetics undergoing surgery on their lower limbs hastened patients' post-surgical recovery.
The doctor's actions were in clear violation of medical ethics. Given that injecting an anesthetic containing preservatives into the sterile, nerve-filled spinal canal may cause neural damage and lead to paralysis, they also demonstrated a complete lack of regard for patient safety.
Example 2: A September 2007 story on the front page of Apple Daily told of a 25-year-old music composer named You Yan-hsiang who had participated in 13 drug studies in a single year, including studies on antidepressants, anticoagulants, and a hepatitis B treatment. A carrier of the hepatitis B virus (HBV), You developed liver cancer as a result of his participation in the studies. Already in Stage 2 when the cancer was discovered, You now regrets trading his health for the NT$100,000 or so he earned from his participation in the drug trials.
You isn't an entirely innocent victim, however. He deliberately hid that he was an HVB carrier and that his family had a history of liver cancer. He also got around questions raised by the abnormal results of his blood tests by claiming that he was taking cold medication. Nonetheless, his ability to participate in so many trials over such a short period at several hospitals and biomedical research centers suggests that the medical community is not terribly conscientious about checking out the subjects of its tests. That being the case, can such trials produce reliable data?
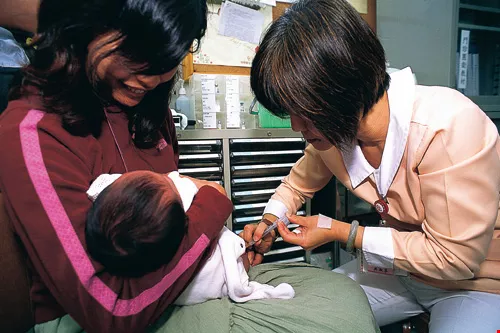
Hepatitis B vaccinations have provided a model for successfully stopping the spread of this disease in Taiwan, greatly lowering the occurrence of the disease and making Taiwan effectively one of the world's largest, most successful "human tests."
A duty or a disgrace?
There are four major types of human trials-healthy-subject studies, diseased-subject studies, diagnostic studies, and treatment trials.
In recent years, Taiwan has been the site of a rapidly growing number of clinical trials, and that just includes the legal ones.
According to the Department of Health's Bureau of Pharmaceutical Affairs, 859 clinical trials are currently under way in Taiwan. Though this figure still lags far behind that of the United States, which leads the world with 27,136 trials, it ranks first in Asia and tenth in the world.
Ironically, however, the draft Clinical Trials Act put forward in 2004 is still languishing in the legislature awaiting review. Under the existing Medical Services Act the Department of Health only has jurisdiction over studies that directly involve the treatment of the ill using new medications, equipment and technologies, with the main focus on pharmaceuticals.
According to the Bureau of Pharmaceutical Affairs, there were 133 applications for human trials of new drugs in 2006, more than 80% of which (110) were developed abroad. Why are foreign drug companies coming to Taiwan to conduct clinical trials? Primarily as a result of market trends and industry demand. "Asia is an emerging market for pharmaceuticals," says bureau director-general Liao Chi-chou. He explains that East Asian nations such as China, Indonesia, Vietnam, Thailand, and Malaysia spend far less on pharmaceuticals than Europe and the United States, suggesting that there is far more room for growth here than in the West. Drug developers are therefore aggressively pursuing clinical trials in Asia.
"These studies are an international duty, as well as something the industry needs," argues Hsueh Jui-yuan, director of the DOH's Bureau of Medical Affairs. Hsueh says that Taiwanese cannot forever sit back and enjoy the fruits of other people's labor, drawing on European and American research while stinting on our own. Though human testing poses risks, it also helps us more quickly learn whether drugs are safe and effective for Asians. As a member of the international community, Taiwan ought to be more proactive about such testing.
In addition, actively pursuing clinical trials would help Taiwan further develop its own biotech industry. "We cannot continue to wait for other people to do the research," says Hsueh. "Or wait for intellectual property protections on new drugs to expire so we can produce them cheaply."
The DOH has therefore been working since 1999 to improve the standard of clinical testing in Taiwan, encourage more transnational drug companies to establish their Asia-Pacific clinical testing centers here, and bring in more advanced pharmaceuticals research and development. Over the last eight years, it has helped 20 hospitals establish what it calls General Clinical Research Centers to encourage medical professionals and drug companies to work together on drug trials.
Some people disagree with the DOH's approach. Former Bureau of Pharmaceutical Affairs director Wang Hui-po wrote an open letter entitled "Are We All Lab Rats?" objecting to government policy's pro-industry bias. Others have asked whether encouraging human trials isn't simply an instance of gambling human lives for economic gain. Such people view Taiwan's ambition to use clinical trials to develop new industries as disgraceful.

Medicine can be a life-saver, but it can also poison and damage the body. As our shelves fill with medicines and drugs, both human tests and follow-up must be carefully monitored. This photo shows the dispensary at Chang Gung Memorial Hospital.
Caged lab rats
Do clinical trials really treat subjects as lab rats? People fear so for good reason.
"Human beings have been doing medical research for a very long time," says Yang Hsiu-i, an associate professor with National Yang-Ming University's Institute of Public Health. "But strict oversight and control mechanisms have only existed for the last 50 years or so." Yang says that up until 1969, before the concept of universal human rights was firmly established, 85% of the world's new drugs were tested primarily on criminals, who were offered reductions to their prison time, or on impoverished victims of disease, who were offered free medical treatment or funeral assistance.
Human testing isn't always about drug trials. Researchers have also been very interested in issues such as healthy subjects' physiological responses to extreme environments, and the pathogenesis and progress of diseases.
During World War II, prisoners of war were the preferred subjects for human experimentation. For example, 23 Nazi doctors used Allied POWs to evaluate human tolerances for extremely high and low temperatures, monitor the progress of fatal diseases, test surgical sterilization procedures, and check the toxicity of drugs, killing or permanently disabling many.
Japanese doctors also put POWs through cruel tests in northeastern China during WWII. There, the researchers of Unit 731 deliberately infected subjects, tested unorthodox treatments on them, and vivisected them. They also tested how much electricity, air pressure, intravenous air, natural gas, and other agents the human body could endure.
The othering of enemies in wartime is destructive to our humanity. But even more shameful to the medical community are doctors who ignore patients' rights in the pursuit of knowledge that may save others. Unfortunately, such things happen.
In New York in the 1960s, researchers seeking to understand how a weakened immune system affected the spread of cancer injected mentally deficient, chronically ill, and immune-compromised patients with cancer cells without their consent. Researchers looking for an alternative treatment for streptococcus infection deliberately withheld penicillin from patients without their knowledge, ultimately causing 25 to develop rheumatic fever.
In 1972, reports began to emerge about a US Public Health Services study of syphilis. Some 400 low-income African-American men known to be infected with syphilis had been tracked for 40 years by researchers seeking to understand the natural course of the disease. The researchers hid the subjects' disease from them, and withheld treatment even after penicillin was revealed to be effective against the condition. Known as the Tuskegee Study, the research ultimately resulted in the deaths of at least 100 people from syphilis or its complications, the infection of 40 of the men's wives, and the contraction of congenital syphilis by 19 of their children, making it one of the most notorious scandals in medical history.
Legal constraints
Damaging pseudo-scientific human testing has so often been carried out in the guise of being "beneficial to society" that people are naturally suspicious.
But the medical field truly is vast, and there is much we still don't know. When developing new treatments, researchers must ultimately-no matter how many pharmacological, toxicological, and animal tests they have run-test them on people to find out if they work. Fortunately, most doctors conducting clinical trials have their patients' interests at heart and are concerned first and foremost for their patients' welfare.
"Doctors with a research bent and an interest in advancing science who are not focused on making a name for themselves will put patients' rights first," says Dr. Lin Kai-hsin, who specializes in pediatric hematological oncology in the Department of Pediatrics at National Taiwan University Hospital. Lin himself is tirelessly seeking new treatments and new medications to relieve the suffering of sick children.
Lin was the first person in Taiwan to conduct human studies on bone marrow transplantation and cord blood transfusions. When he carried out Taiwan's first sibling bone-marrow transplant in 1984, Taiwan had no legal guidelines in place covering this kind of procedure.
Bone marrow transplants and cord-blood transfusions have since become standard treatments. These days, some doctors, feeling they must keep up with the times, go ahead with transplants when the DNA samples show a tissue match for as few as a four out of six markers. While not illegal, such a decision by the doctor is tantamount to homicide.
Great care must be exercised when carrying out invasive medical procedures such as surgery. Even though drug testing is a common practice and seems safer than surgery, a similar degree of caution is required.
Generally speaking, clinical drug trials are divided into four phases.
The first phase of human trials follows the completion of animal testing. Its objective is to understand the drug's toxicity in humans, and looks at biochemical and physiological phenomena such as absorption, distribution, metabolism, excretion, and tolerability. Phase 1 trials are usually conducted on healthy subjects because the skewed physiological responses of ill subjects would yield unreliable data.
Phase 2 trials include a small number of ill patients, and test the drug's effectiveness, toxicity, safety, and tolerability at various dosages. The trials seek to find appropriate dosage levels, and typically involve the participation of several medical centers.
Phase 3 trials provide the definitive assessment of a drug's effectiveness. Typically large and multinational, these trials are conducted on patients with the relevant disease. Phase 4 trials take place after the drug has been released to the market. Their objective is to monitor for the appearance of previously hidden adverse reactions and side effects.
Currently, a relatively low proportion of Taiwan's clinical trials are the more dangerous Phase 1 trials. In fact, only 12 of the 133 new drug trials approved by the DOH in 2006 were Phase 1 trials. In addition, only one of the 92 hospitals approved for human trials by the Joint Institutional Review Board carried out Phase 1 trials in 2006.
Liao Chi-chou says that the reason Taiwan carries out so few Phase 1 trials is that Phase 1 trials are very small. Because they typically involve only 20-80 people, most drug companies simply carry them out wherever they happen to have developed the drug.
Others argue that it is the legal system that keeps the number of Phase 1 trials low.
In contrast to the Anglo-American legal system's strong reliance on precedent in judging cases, Taiwan, like mainland Europe, has a "civil-law" legal system, in which judges focus more on the wording of the statutes themselves. Though many of the provisions of the relevant legislation are rarely applied, the prospect of landing behind bars as a result of an oversight in the treatment of a patient or during a human trial makes doctors very nervous about working with drug companies on Phase 1 trials, which are filled with unknowns.
"What's there to be afraid of!" exclaims Hsueh Jui-yuan. He says that though clinical trials have yet to be "decriminalized," no researchers to date have been prosecuted or even investigated. As long as they follow all the regulations, they have nothing to fear. "On the other hand, those who try to avoid hassles by doing things cheaply or secretly in violation of the law will face severe legal sanctions should anything go wrong."
Who are the lab rats?
Phase 1 trials may carry greater risks, but they represent a crucial first step in human trials.
"From a scientific standpoint, if you are going to do world-class work, it is crucial to make the transition from the laboratory to the clinic," says Chang Tse-wen, a research fellow in genomics at the Academia Sinica. According to Chang, who co-founded an American pharmaceuticals company and developed Xolair, a drug used in treating allergic asthma, the doctors who win the favor of the pharmaceuticals companies and conduct Phase 1 trials are always those at the top of their field-those who have the greatest expertise, have contributed the most and have won the highest honors. But the regulatory regime in Taiwan has led the medical community to view this field as being off limits.
"If the whole world were as 'selfish' as Taiwan," says Chang, "there'd be no way to develop new drugs." Chang argues that Taiwan's legislators don't understand drug development. Phase 1 trials are the most crucial, most prestigious and most valuable part of drug research. The fact that Taiwan has to depend on other countries to carry them out is an obstacle to drug development here.
"It's wrong to compare Phase 1 subjects to lab rats," says Chang, who believes that there's something dubious about having someone else undertake trials you are unwilling to undertake yourself, given that equality is one the objects of human rights. This is truer still when you consider that Phase 1 trials are carried out under the close supervision of doctors at the clinical research centers, and that the dosages start at very low levels and rise very slowly to establish a tolerability profile for the drug in healthy subjects. This close supervision makes Phase 1 trials much safer than Phase 2 and 3 trials, in which ill patients administer drugs to themselves in their own homes. Chang therefore argues that participants are nothing like lab rats.
Generally speaking, Phase 2 and Phase 3 trials are less controversial, and patients tend to be very willing to participate.
Drug development is expensive and time consuming. Liao Chi-chou says it takes US$200 million to US$1 billion to develop a new drug and five to 15 years to get the regulatory approval required to bring it to market. Most of the new drugs undergoing clinical trials target diseases and conditions for which there is currently no cure, such as rare diseases, cancers, cardiovascular disease, diabetes, and high blood pressure. If Taiwan were to become involved with clinical trials at an earlier stage, many sick people anxiously awaiting new drugs could benefit.
The testing of drugs to treat AIDS in the 1980s is a case in point. Victims of the disease were eager to participate in drug trials because such trials offered them a chance at life. Once in, subjects used every means they could to find out if they were actually receiving the new drug or a placebo. In some cases, they even resorted to swapping drugs in an effort to improve their odds of surviving.
But healthy people have no such pressing incentive to participate. How do you get them involved in Phase 1 trials?
Chang says that while you can't legally offer "incentives," you are permitted to provide reasonable remuneration and compensation.
In general, drug trials seek healthy men under the age of 25. Participants earn an average of more than NT$10,000 per study (for "nutritional expenses"), which can look pretty good to a university student. Not surprisingly, many end up taking "part-time jobs" participating in a variety of human trials. Students with medical backgrounds are highly sought after because of their understanding of and sensitivity to physiological reactions, and they participate at the highest rates.
September 2007 media reports on "professional drug testers" finally prompted the DOH to publish guidelines on the recruitment of subjects for clinical drug trials. These guidelines strictly prohibit the placing of recruitment notices on the grounds of schools of middle-school-age and younger children, and bar notices from stating or implying that the drug being tested is safe or effective. But they do nothing to prevent the drug companies from providing cash-strapped students with incentives to participate.
Doctor vs. researcher?
Subjects are often drawn to studies by "incentives." What gets doctors involved?
"Study doctors play two roles, in that they are both doctors and researchers," says Liu Hung-en, who teaches law at National Taipei University. "The conflict between the two has never received a lot of attention." Liu says that attending physicians at university hospitals must teach and publish on top of treating patients. As a result, they are always both doctors and researchers. But a doctor's research objectives and a patient's best interests are not always identical, and may even be at odds with one another. In such circumstances, it is hard to guarantee that a doctor will not give a patient a treatment as a pretext for research or even ignore the patient's rights entirely. For example, a doctor might continue an experiment in spite of a patient's bad reaction to it.
Liu also wonders whether the drug companies' funding of research affects the conclusions the doctors and researchers reach.
In How Doctors Think, for example, Dr. Jerome Groopman notes that the medical community has yet to confirm whether men experience a kind of menopause. He states that in spite of little research showing clear benefits from hormone therapy to middle-aged men suffering from low levels of hormones, declining muscular strength, and reduced sexual desire, doctors heavily promote male hormone treatments, thereby generating booming sales of hormone products.
Groopman writes that the drug companies are out to make money, and to that end do their utmost to influence how doctors think, and specifically what diseases they recognize and how they treat them. In Groopman's view, this situation has overstimulated demand for drugs, likely been detrimental to patients' health, and given rise to unnecessary side effects.
"The drug companies own the results of clinical trials," says Dr. Benjamin Kuo of Veterans General Hospital in Taipei. He explains that the companies frequently spend more than NT$100 million on a drug trial and it is they who decide whether to publish the results. They are naturally selective, releasing the positive conclusions and keeping quiet about the negative ones. As a result, the public never gets the full picture.
Another issue is that medical research in areas other than treatment and drug trials often takes place outside of hospitals. Psychological and physiological experiments can severely harm subjects' physical and mental wellbeing, but don't currently fall within the purview of the law.
Hsueh Chi-yuan admits that Taiwan's legal protections for study subjects fail to live up to the spirit of the Helsinki Declaration. Protections, particularly for subjects who are disadvantaged or already receiving medical treatment, will need to be strengthened when the Medical Services Act is revised.
If we are to promote the advancement of medical and pharmacological science, we cannot hobble researchers nor censure them at every turn. In addition, we need subjects willing to risk participation in studies. But, having granted these points, we must always, in every circumstance, demand that researchers respect every single life.


@List.jpg?w=522&h=410&mode=crop&format=webp&quality=80)



