Cancer applications
Lon proteases have attracted interest for their possible application in the fight against cancer.
The effects of some targeted drugs, says Chang, have not yet been narrowed to cancerous cells, but progress might be made by suppressing the ability of cancerous cells to dispose of their refuse.
In theory, if a drug can bond with the six activation domains in the LonC molecule’s central chamber and suppress its capacity to dispose of useless proteins, then the cancerous cell will not be able to proliferate. Current research indicates that Lon proteases are very important to the survival of some kinds of cancerous cells, lymphoma being one example. And some pathogenic bacteria, such as typhoid, also need Lon proteases in order survive within the host.
Some drugs are effective, but the reasons for their efficacy are unknown. Now that the structure of LonC protease has been resolved, however, it is possible to determine how it could be incorporated into a small-molecule drug. This would make it possible to improve drugs by making them even more precisely targeted. A targeted cancer drug called Velcade, for example, was originally used clinically to suppress the breakdown of proteins, but Chang discovered that it also suppresses the activity of mitochondrial Lon proteases. The discovery inspired two new directions in his research. First he began working to improve Velcade by eliminating its suppressant effect upon mitochondrial Lon proteases, so as to narrow the drug’s effect and reduce side effects. Then, building on this work, he set about designing a suppressant for Lon proteases.
“Doing research on a local bacteria is like a dream come true,” exclaims Chang, adding that one of the best things about scientific research is that one study leads to another. For example, LonC protease is ATP-independent, while the LonA protease in human mitochondria and yeasts is ATP-dependent. Chang surmises that perhaps the pore to the chamber in LonA protease is relatively small and therefore needs energy from ATP to get proteins into the chamber, while the pore in LonC protease is larger and therefore needs no ATP. However, he stresses, “the difference between them remains to be proven.”
So, it turns out, the “beauty springs” of Wulai do a lot more than just invigorate the circulation and beautify the skin. Now we find that bacteria there harbor secrets awaiting scientific discovery. And some scientists from Taiwan have already begun to crack the code!
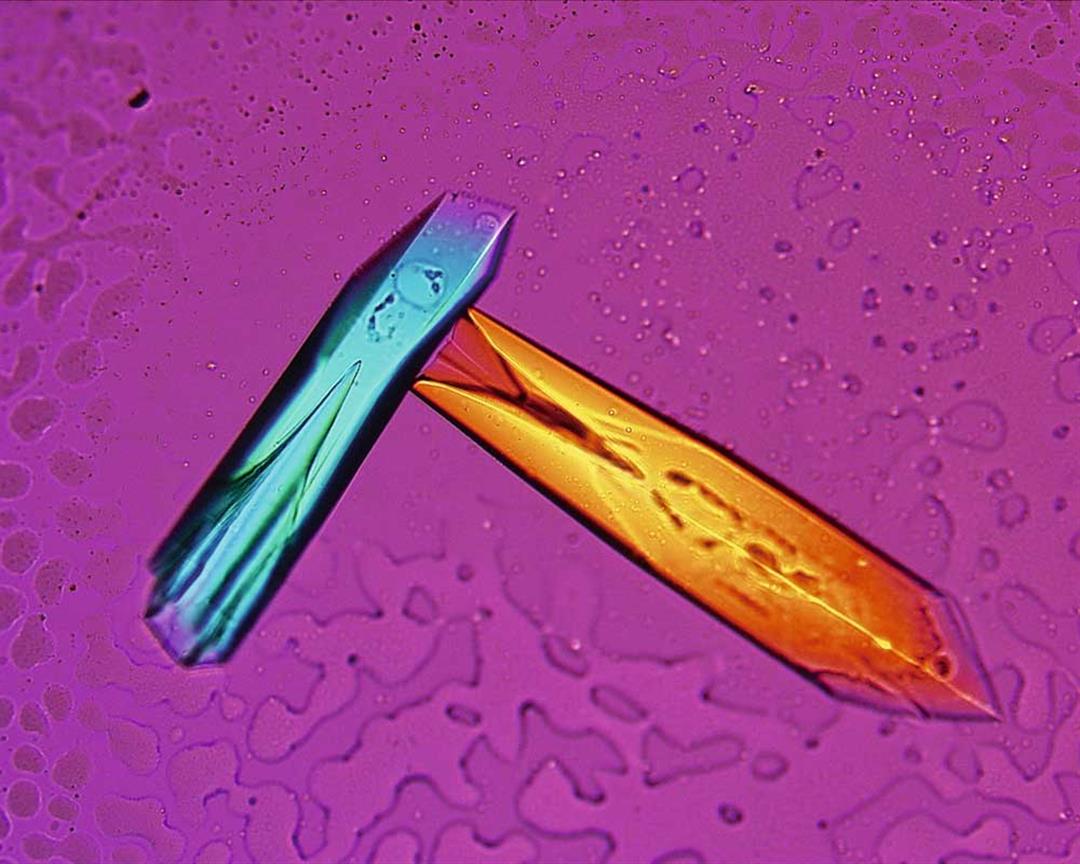
The refracted light from a laser beam passed through a protein crystal presents a beautiful image.
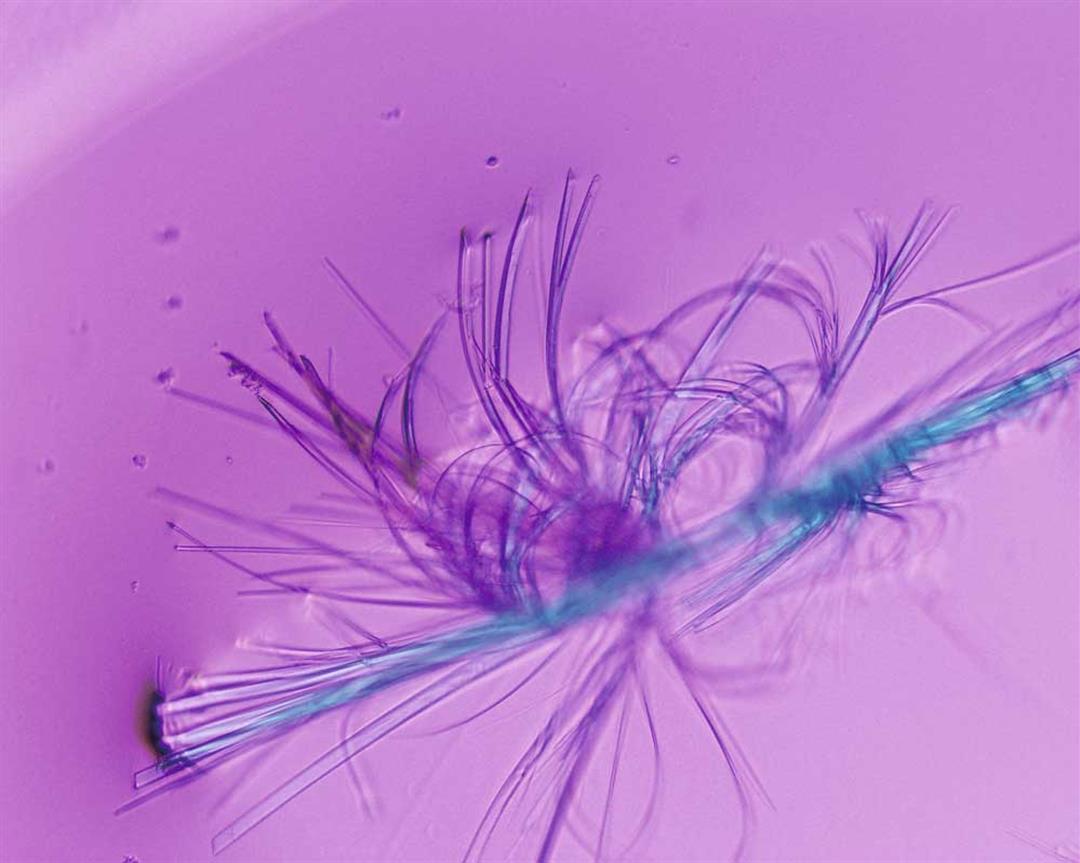
The refracted light from a laser beam passed through a protein crystal presents a beautiful image.
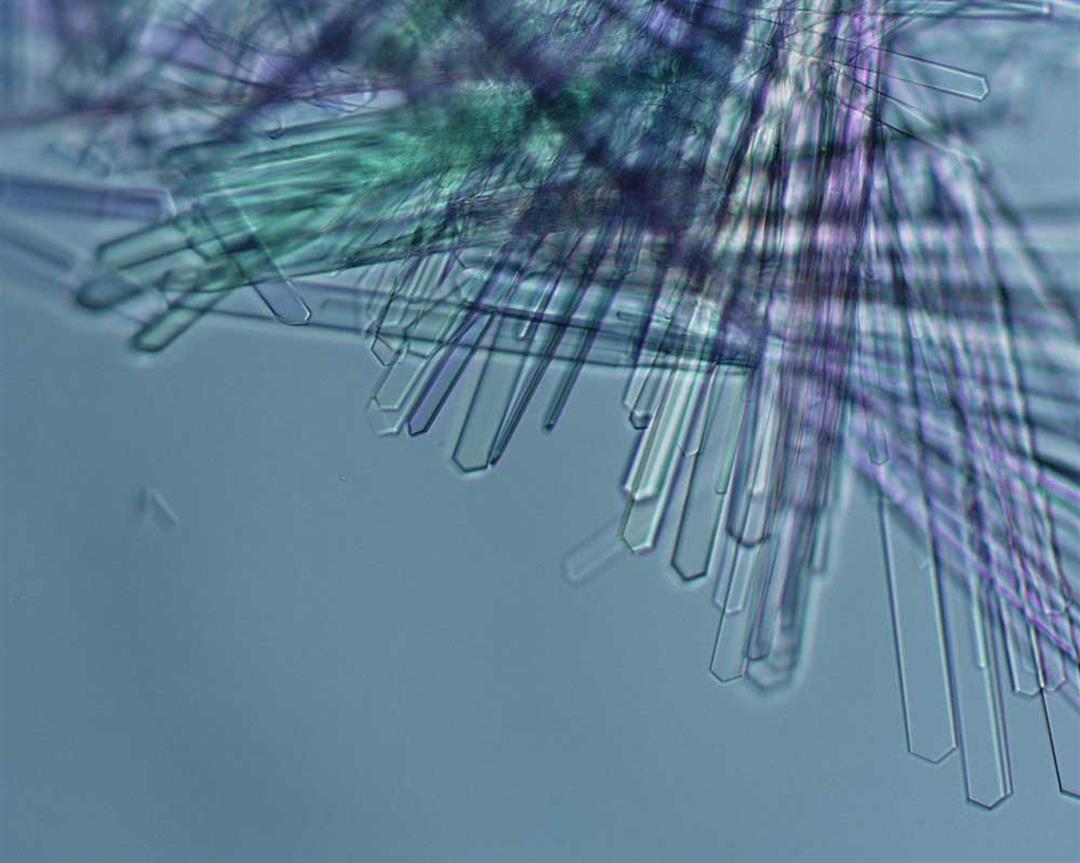
The refracted light from a laser beam passed through a protein crystal presents a beautiful image.

The raw data obtained through X-ray crystallography requires further computational analysis.
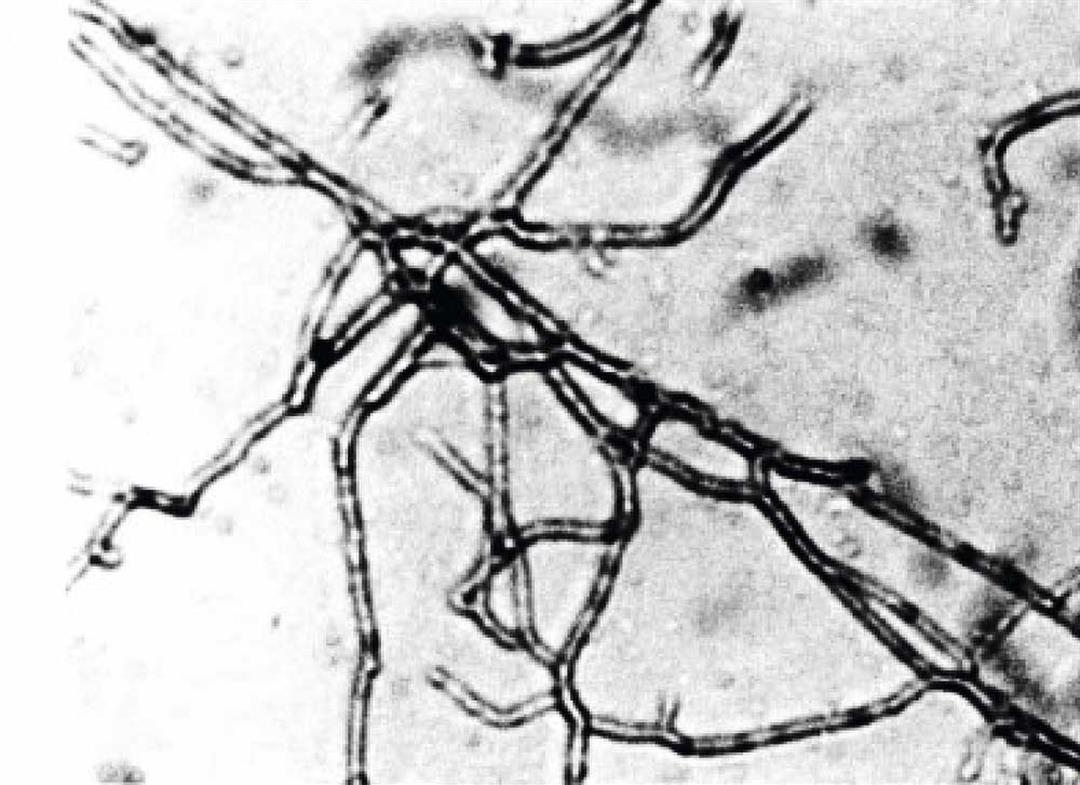
Under the microscope, a thermophilic bacterium discovered in the hot springs of Wulai, south of Taipei.Its discoverers have named it Meiothermus taiwanensis.

The raw data obtained through X-ray crystallography requires further computational analysis.