Tears from Heaven--Taiwan's Acid Rain Crisis
Lin Hsin-ching / photos Jimmy Lin / tr. by Scott Williams
December 2009

In Chinese we often describe impos-sibilities as "grain from the sky and horses with horns," or just "raining rice." But nowadays Taiwan often sees acid from the sky. In fact, more than 57% of the rain that falls on Taiwan now has a pH of 5.0 or less, the official threshold for acid rain. The problem is most severe in northern Taiwan, where 80% of the rain that falls meets the definition of acid rain. Areas such as Keelung and the Yangmingshan saddle frequently see rains and fogs with a pH of just 3.0 or so, a level nearly equivalent to the roughly 2.9 pH of vinegar.
The effects of acid rain aren't limited to possible balding in those exposed to it. Research both in Taiwan and abroad has shown that the effects of acid rain accumulate over time. In fact, acid rain has already damaged river, lake, and forest ecosystems, agricultural harvests, architectural structures, and many people's health. According to estimates from Taiwan's Environmental Protection Administration (EPA), acid rain causes annual economic losses in excess of NT$10 billion. Taiwan is clearly in urgent need of a solution to the environmental problem of acid rain.
On October 2, the eve of this year's Mid-Autumn Festival, just as Typhoon Parma was approaching Taiwan, the northeast monsoon rolled in, showering northern Taiwan with intermittent rains. Though the windward side of the Yangmingshan saddle received only nine millimeters of rain, that rain had a pH of 3.2. In other words, the rain that fell on Yangmingshan that day was more acidic than either tomato juice (4.2) or orange juice (3.5).
"The rainfall we receive when the northeast monsoon strengthens and from the leading edge of frontal systems is especially acidic," says Mr. Xie, an employee of the Central Weather Bureau's Yangmingshan weather station. "It's only after a couple of days, when the air has been washed clean of pollutants, that conditions improve."

Attacked by acid rain over the years, many of the marble statues on Rome's streets have become darkened and disfigured, obscuring their richly expressive facial features.
A product of pollution
The idea of orange- or tomato-juice rains sounds more than a little ridiculous, but equivalent levels of acidity are common in the rains falling on northern Taiwan in the autumn and winter. Why is our rainfall so acidic? To answer that, we must first look into the causes of acid rain.
Lin Neng-huei, a professor in the Department of Atmospheric Sciences at National Central University and head of the EPA's acid rain measurement project, says that chemistry defines a pH of seven as neutral, and less than seven as acidic. Our atmosphere contains a great deal of carbon dioxide, which when dissolved in water gives us slightly acidic rain (pH of roughly 5.6). Then there are natural acidifying elements, like the hydrogen sulfide belched out by volcanic eruptions, the dimethyl sulfide emitted by the oceans, the nitrogen oxides created by lightning at high altitude, and even the various sulfur and phosphorus compounds produced by forest fires. Collectively, these can bring the pH of rain down to 5.0 or so.
"Rainfall in the natural world is already slightly acidic. That's normal," explains Lin. "But when the pH of rainfall drops to less than 5.0, that's definitely an unnatural, manmade phenomenon. And that's the definition of acid rain."
Lin says that the scholarly community generally agrees that manmade pollutants such as sulfur oxides and nitrogen oxides are the primary causes of the dramatic acidification of rain worldwide. The former are suspended particulates largely produced by factories or power plants burning coal or other fossil fuels. The latter originate primarily in automobile exhaust. When dissolved in water, these anthropogenic pollutants become sulfuric acid and nitric acid, furthering the acidification of rainwater.
Rain with a pH of less than 5.0 is an anthropogenic pollutant, a product of modern industrialized society. There is no controversy on this point. But processes such as dry deposition, in which suspended particulates and acid-forming substances in the atmosphere are often blown to other locales, and wet deposition, in which these substances bond with cloud droplets then fall as rain, snow, fog, or hail, mean that acid rain is always somebody else's problem. That is, areas suffering from acid rain are never the main culprits in its production. Instead, they tend to be the victims of pollution that originates in other locales or even other industrialized nations. Taiwan provides an excellent case in point.
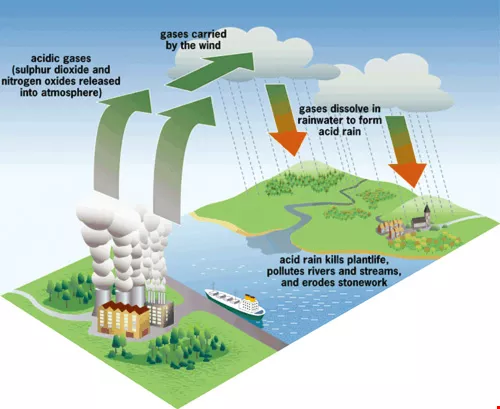
In the age of the "global village," the wind carries all kinds of pollutants across the sea where it ends up falling on people uninvolved in its manufacture. Acid rain is a case in point. It is a serious transnational ecological issue.
Across the sea
An EPA survey covering 1991-2008 showed that 57% of the rain that fell in Taiwan was acid rain. Acid rain was more common in the north, and most severe on Pengjia Islet, Keelung, the Yangmingshan saddle, Taipei, and Zhongli in Taoyuan County, in which locations its incidence was in excess of 80%. Seasonally speaking, acid rain is most common in fall, winter, and spring. During the December to February period, its incidence rises as high as 71% for Taiwan as a whole while the average pH falls to 4.76.
The acidity isn't as severe from May to September, when the pH of rainfalls across Taiwan tends to stay above 5.0.
Why does acidity vary with the season? In Taiwan, it's because more than half of our acid rain is caused by pollution from outside our borders. Pollution from "the world's factory," mainland China, is the most direct source. When the northeastern monsoon intensifies in fall and winter, fronts blow in from northern China, picking up industrial emissions of sulfurous particulates from northern, central, and southern China as they blow south. These eventually reach Taiwan, which is downwind in these seasons.
"When weather systems carrying acidic particulates encounter elevated terrain or warm southern air, water condenses and acid rain results," says Peng Chi-ming, one of the leaders of the EPA's acid rain monitoring program and CEO of Weather Risk Explore. "Northern and northeastern Taiwan get the worst of it."
But Taiwan can also experience abnormal "alkaline rain" during the fall-spring period. This usually occurs when the EPA has issued dust-storm warnings. Peng says that winter and spring are the dust-storm season in northern China. Because the principal components of this dust are particles of alkaline substances such as calcium and magnesium, which neutralize the acidic particulates in rainwater, dust storms can lead to "alkaline rains" with a pH of 7.0 or higher in Taiwan.
During the summer, Taiwan's rains are primarily influenced by afternoon convection, the southwest monsoon, and Western Pacific typhoons. These weather systems less frequently pass through areas of industrial pollution, so the rain tends to be cleaner and have a higher pH.

The clouds and rains that blanket luxurious mountain residences give them a romantic air. Unfortunately, these misty, artful scenes are often painted in pollution-laden acid fogs that represent a threat to forests, buildings, and human health.
Foreign and domestic culprits
Taiwan's own heavy industries and power plants also create a variety of sulfurous particulates. These, together with the exhaust from ever-increasing numbers of automobiles, are domestic accomplices in the production of acid rain.
Taiwan experienced its most severe acid rain problems in the 1990s. Measuring instruments in Taipei and Kaohsiung revealed average rainfall pH values of 4.46 and 4.81, respectively, in the two metropolises. But in 1995, the EPA began levying new air pollution prevention fees on fixed sources of pollution (e.g. factories, power plants, and construction projects) and mobile sources of pollution (e.g. cars and motorcycles). The fees target businesses' emissions of sulfur- and nitrogen oxides, and have led to a gradual increase in the pH of rainfall in the two metropolitan areas. Since 2000, the average pH of rainfall in Kaohsiung has risen to more than 5.0, while in Taipei it has increased to 4.6-4.7.
To exercise greater control over Taiwan's sulfurous particulate emissions, the EPA plans to lower the limit on the sulfur content of diesel fuel from its current level of 50 parts per million to just 10 ppm in 2011, and to manage the sulfur content of gasoline in the same manner beginning in 2012. The EPA also plans to reduce the permissible sulfur concentration of industrial fuels from its present level of 0.5% to 0.3%, but is still discussing the timeframe for implementation with industry.
But the government's tighter standards on sulfurous particulate emissions have yet to yield much improvement in northern Taiwan's acid rain problem. According to Wu Cheng-tao, a director with the EPA's Department of Air Quality Protection and Noise Control, there are two reasons for this. First, domestic reductions in sulfur oxides haven't matched the nearly 40% per year rise in Chinese emissions. Second, the rapid growth in the number of automobiles on the road in Taiwan (from 16.3 million motor vehicles in 1999 to 21.2 million in 2009) has resulted in increased domestic nitrogen oxide emissions. Acid rain therefore continues to be a problem.

State-to-state talks
Taiwan isn't the only place to suffer from acid rain pollutants produced elsewhere. In the 1970s, Norway and other Scandinavian nations discovered that their lakes were becoming more acidic (a healthy lake has a pH of 8.0-9.0) and that fish populations in those lakes were shrinking. Studies eventually discovered acidic pollutants in the local rain that had been produced by heavy industry 1,500 kilometers away in northern Germany's Ruhr region. Acid rain was also killing large numbers of trees in southwestern Germany's Black Forest. A West German government study found that prior to 1983 some 34% of the nation's 7.4 million hectares of forest had died as a result of acid rain, and that industrial regions in the northern and central parts of the United Kingdom were the likely culprits.
In the 1980s, the US and Canada discovered their own acid-rain damage. Acidic particulates emitted by heavy industry on the US side of the Great Lakes were being blown elsewhere, killing large numbers of trees in the forests on the western slopes of the Appalachian Mountains and acidifying Canadian lakes near the US-Canadian border. When the pH of these lakes fell to less than 5.0, they became fishless "dead lakes."
The situation didn't improve until the US began implementing a national acid-rain-prevention program in the 1980s and 1990s that required power plants to take measures to control their total sulfur emissions. But even with these measures, the ecological wounds already inflicted have yet to heal.
It is exceedingly difficult to quantify pollution that comes from beyond one's borders. The Canadian government-the self-designated victim-repeatedly sought compensation from the US, but the two countries were never able to arrive at an agreement. Neither has any European nation been successful in its efforts to seek recompense, and acid rain remains a very difficult ecological issue in transnational negotiations.
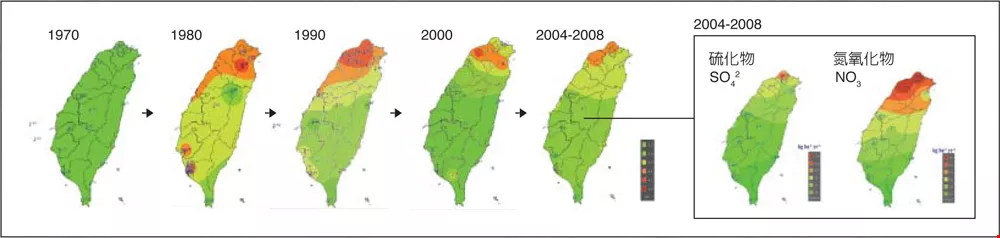
The series of images at left above, showing the distribution of acid rain in Taiwan from 1970 to 2008, demonstrates that years of diligent effort have improved the acid rain situation. The two images at right show an analysis of the constituents of acid rain from 2004 to 2008. They show that sulfurous particulate concentrations are most severe on the windward side of high elevations in northern Taiwan, while nitrogen oxides are more widely distributed. This indicates that not only are pollutants from outside Taiwan a problem, but that steadily increasing domestic tailpipe emissions are also an issue.
An ecological killer
Why does acid rain, sometimes referred to as "the Grim Reaper of the atmosphere," wreak such irreparable ecological harm around the world?
Lin Teng-chiu, a professor in the Department of Life Science at National Taiwan Normal University who has studied acid rain ecology, says that while acid rain is perhaps not the principal culprit in large-scale forest die-offs, it is an important accomplice. Acid rain contains a huge numbers of hydrogen ions, which are positively charged and highly reactive. When these enter the ecosystem, they displace calcium, magnesium, and potassium ions. The displaced minerals then bond with negatively charged sulfate ions and are washed out of the ecosystem.
The chemical effect of these ion exchanges is to leach nutrients out of soils in these ecosystems, inhibiting plant growth. When plants lack calcium, newly sprouted leaves and branches die easily. When they lack magnesium, they have a hard time synthesizing chlorophyll and carrying out photosynthesis to produce the nutrients they need to grow, and become much less resistant to blight.
Nitrogen is another factor affecting ecological systems. Nitrogen is an important component of proteins and is essential for plant growth. But when acid rain carries the nitrogen oxides emitted in automobile exhaust into the ecosystem, the normally nitrogen-deficient natural world becomes saturated with it.
"In the past, trees would extend their roots deep into the earth to get a little more nitrogen to help themselves grow," says Lin. "But now nitrogen is too easy to come by. Plants don't need to expend the energy to put down stable root systems. As a result, trees are now easily uprooted by typhoons and other storms."
In addition, excess nitrogen in the ecosystem causes plants to overgrow. This makes them especially attractive to pests, which represents yet another danger.
Lin says it is possible that the use of lime and other human interventions can facilitate normal plant growth on agricultural lands affected by acid rain by adjusting the pH of the soil. But there's almost nothing humanity can do about vast forests and lakes. "As with the tragic decline in the European and North American forests, despite all the efforts to save them, ultimately all you can do is watch helplessly as it plays out."
In addition to causing ecological harm, acid rain can also damage buildings and sculptures, especially those made from sandstone, limestone, or marble. Acid rain eats into such materials, and, through a series of chemical reactions, turns the hard calcium carbonate on their surfaces into powdery white calcium sulfate (gypsum), which then cracks and flakes off. In Athens, years of acid rain erosion have fouled the faces and garments of many of the sculptures of goddesses on the Acropolis.
But it isn't only historical sites that are suffering: acid rain can eat into modern buildings as well. Lin Neng-huei, the head of the EPA's acid rain monitoring project, teaches at National Central University. The university happens to be located in Zhongli, Taoyuan County, which has a severe acid-rain problem. Lin says that when he first came back to Taiwan to teach more than a decade ago, the brick red exteriors of the College of Science's just-completed Science Buildings 2, 3, and 4 were beautiful to look at. But after a few years the windward sides of the buildings began turning black and some of the bricks began cracking and falling, posing a serious threat to the safety of faculty and students.
To prevent the threat from worsening, the school ended up budgeting a large amount of money to rebuild the exterior walls. "Northern Taiwan's rainy, humid climate by itself shortens the useful lives of buildings," says Lin. "Acid rain just makes matters that much worse."

Acid rain can also damage modern buildings. National Central University is spending big money to give a group of buildings belonging to the College of Science a "face lift" to repair their cracking and flaking exteriors, damage wrought by acid rain.
Allergies and respiratory disease
But what about the health effects of acid rain? Dr. Lin Ja-liang, head of toxicology at Linkou's Chang Gung Memorial Hospital, says that most people think acid rain's health effects are limited to hair loss, but this is only half true. The acidic materials in acid rain can indeed irritate skin, making immune responses such as allergic itching and inflammation more severe. If this inflammation occurs on the scalp, it can damage the skin and even lead to hair loss.
"But in reality few people, other than adolescents who can't be bothered, fail to use an umbrella when it rains," says Lin. "Consequently, acid raid does relatively little direct harm to people's health."
Of more concern is that areas where acid rain is more frequent typically have higher levels of atmospheric sulfur oxides and nitrogen oxides. The former can cause asthma and inflammation in the trachea and bronchioles, while the latter breaks down in sunlight to form ozone, a powerful oxidant that is both toxic and very reactive. Ozone not only attacks and inflames the respiratory tract, but also impairs the lungs' ability to exchange gasses, reducing oxygen levels in the blood. In severe cases, it can even lead to emphysema or other forms of severe lung damage.
"Whether it rains or not, on days when atmospheric concentrations of sulfur oxides or nitrogen oxides are high, the elderly, children, and people who suffer from cardiovascular disease should avoid going outdoors," says Lin. "And if they really must go out, they should wear a mask to reduce the damage."
Scholars also remind people to be wary of "acid fog." Because the water droplets in fog are smaller, the acid in them tends to be more concentrated and of a lower pH.
London's Great Smog of December 1952 was caused by the carbon monoxide, sulfur oxides, and soot released by the burning of large amounts of coal within the city. A series of several windless days allowed the pollutants to accumulate and with the fog droplets to form a toxic smog with a pH of just 1.6, a level that made it almost as corrosive as sulfuric acid. The smog resulted in severe respiratory problems for thousands of Londoners, killing some 4,000 people over just four days and another 8,000-plus over the following two months.
A "romantic" crisis
Though Taiwan hasn't experienced such a terrible acid fog, years of surveys have shown that acid fogs in the mountains really are much more acidic than acid rains. For example, Lin Neng-huei's research team has gathered samples of acid fog in the Yangmingshan area with a pH of just 2.6. (For comparison, Coca-Cola has a pH of 2.5.) And Lin Teng-chiu's research team has similarly collected samples of acid fog with a pH of 3.8 in the mountainous Xitou area of Nantou County, a popular tourist destination.
Lin Neng-huei, Lin Teng-chiu, and Peng Chi-ming all have much the same thing to say about acid fog: While we naturally use umbrellas when it rains, umbrellas are useless in fogs, which surround us and soak us through. Since there's no easy way for us to protect ourselves from fogs, the scientists argue that acid fogs are a greater worry than acid rain.
Lin Teng-chiu adds that behind many a romantic scene lies a good deal of pollution-e.g. flame-red sunsets (dust scattering sunlight makes sunsets redder), green mountain lakes (a symptom of severe eutrophication), and lingering clouds and mists.
"Maybe we should update the old saying 'sunset is beautiful but the night is coming' to 'sunset is beautiful but the pollution is heavy,'" remarks Lin Teng-chiu wryly.
Gathering long-term data
Extrapolating from the experience of other nations, Lin Teng-chiu and others have shown that acid-rain damage to Taiwan's ecology, agriculture, architecture, and human health results in total economic losses of as much as NT$10 billion per year (roughly 0.16% of GDP).
But, as serious as this damage is, scholars and EPA officials admit that there's very little that Taiwan can do about it since most of the pollution originates in mainland China. The EPA has sought to bring up the problem of long-range transmission of pollutants (e.g acid rain) at several recent Cross-Strait Economic and Trade Forums, but the Chinese negotiators have avoided the topic, and instead agreed only to include weather cooperation in the negotiations.
"The reason is simple," says an exasperated Lin Neng-huei. "Weather cooperation benefits people's livelihoods, while pollution prevention requires determining blame. Japan has been trying without success to bring China to the negotiating table for years. Taiwan will have an even more difficult time doing so."
If we are to address this problem in the long term, we can and must improve our own pollution controls, collect long-term monitoring data, and work closely with acid-rain monitoring networks in other East Asia nations. With regard to China, which truly is the elephant in the room on this issue, all we can do is prepare, wait for an opportune moment, then, with the help and support of the international community, encourage it to address its various pollution issues.

@List.jpg?w=522&h=410&mode=crop&format=webp&quality=80)





